AgriLife Research to investigate ‘mechanics’ of botanical compounds and human microbiota
Study looks at science-based recommendations for dietary phenols for reducing intestinal inflammation
Texas A&M AgriLife Research has received a $632,593 one-year grant from the National Center for Complementary and Integrative Health, NCCIH, for a study related to complementary and alternative medicine.

One of the principal investigators for the study, called ’Anti-inflammatory microbiome-substrate-host interactions,’ will be Susanne Talcott, Ph.D., AgriLife Research associate professor in the Department of Food Science and Technology in the Texas A&M College of Agriculture and Life Sciences, Bryan-College Station. She will collaborate with Robert Britton, Ph.D., Baylor College of Medicine in Houston, and Stephen Talcott, Ph.D., also in Texas A&M’s Department of Food Science and Technology.
“This is a high-priority, short-term project,” Talcott said. “The National Institutes of Health has invested millions into the clinical investigation of botanical compounds, especially dietary phenols. However, to date, most human clinical intervention studies of these compounds have failed to provide consistent results.”
She said age and obesity are associated with a lower ability of the intestinal microbiome to produce absorbable metabolites from polyphenols.
“This means that the very population in need of the anti-inflammatory properties of fruits and vegetables may not optimally benefit,” she said.
The NCCIH is part of the National Institutes of Health, NIH, in the U.S. Department of Health and Human Services. Talcott said the one-year grant is a bridge toward a larger, still pending, application for almost $4 million in funding.
What are polyphenols?
Polyphenols are a set of healthful substances present in fruits, green tea, spices, nuts and other foods. The biggest subset of polyphenols people ingest from foods are substances called gallotannins. People’s intestinal microbiomes vary and process polyphenols differently, and emerging evidence suggests that this fact has contributed significantly to inconclusive human clinical trials with polyphenolics.
“Studies have shown that microbial metabolites have anti-inflammatory effects through cell signaling, but only in those individuals who actually have bacteria in their intestinal microbiome that can break down gallotannins into smaller absorbable anti-inflammatory metabolites,” Talcott explained.
She said a lack of understanding of the causes of the person-to-person variability underlying the efficacy of natural products is burdening human clinical studies that evaluate the biological signatures indicative of improved resilience to chronic diseases.
“The NCCIH is seeking to better understand if and how these botanical compounds may be effective in reducing inflammation,” she said.
The purpose of the study
Talcott said the long-term goal of this new project is to help develop novel recommendations for safe and effective consumption of large molecular polyphenols. This includes looking at gallotannins and other anti-inflammatory polyphenols within a precision-nutrition approach to resilience against inflammation.
“Gallotannins themselves are not metabolized directly by humans but are substrates for select bacteria in the human gastrointestinal tract,” Talcott explained. “Metabolism of gallotannins generates bioavailable anti-inflammatory metabolites, but concentrations of these metabolites show large variation between individuals.”
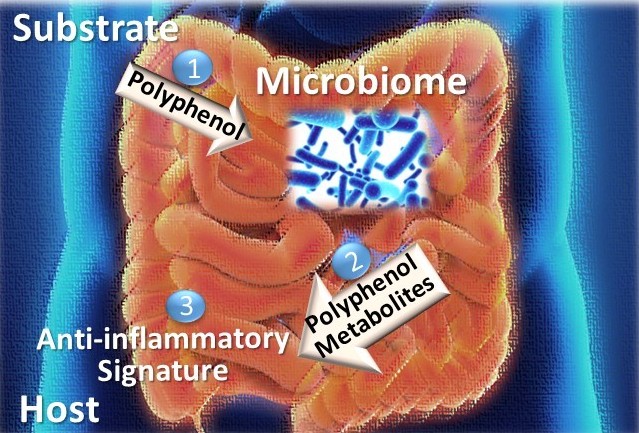
She said polyphenol metabolism may be limited in individuals with chronic intestinal inflammatory conditions, which might be addressed via supplementation with targeted probiotics.
“Two well-defined intestinal microbial enzymatic steps yield the major bioactive metabolites of gallotannins,” she said. “These are tannase and tannin acyl hydrolase GA-decarboxylase.”
She said both enzymes are active in the Lactobacillus plantarumstrain and present a suitable model to study gallotannin metabolism.
“We are hoping to define the underlying microbial molecular basis for the inter-individual variability in the intestinal anti-inflammatory effects of gallotannins.”
She said the study will develop a proof-of-principle model using knock-out versions of the Lactobacillus plantarumstrain that naturally encodes both enzymes and presents an optimal model for gallotannin metabolism.
“Our central hypothesis is that inter-individual differential microbial metabolism of gallotannins is a significant determinant of their anti-inflammatory activities in regard to intestinal inflammation,” she said.
Talcott said finding a mechanistic determination of how substrate-microbiome-host interactions influence the anti-inflammatory activities of gallotannins will enable the development of more formally science-based intake recommendations or dosages for botanical polyphenols.
“Principles from this study can be transferred to human clinical studies and further the scientific basis for dosing recommendations for gallotannins and other polyphenols in increasing resilience against chronic inflammatory diseases,” she said.
Growth in use of botanical supplements
The NIH estimates the intake of dietary botanical supplements has been steadily increasing and was a $7 billion industry in 2018, despite a lack of scientifically based intake recommendations. At the same time, the incidence of inflammatory bowel disease, including Crohn’s disease, and ulcerative colitis affected 1.6 million people in the same year.
The global botanical market is projected to exceed $49 billion by 2027, with many of the supplements based on a polyphenolic bioactive, despite an absence of human clinical evidence showing consistent pharmacokinetics and efficacy in the prevention of a wide array of ailments, including intestinal inflammation.
“Where standard medications are associated with severe side effects and there is poor medication adherence, many patients seek alternative botanical remedies, such as prebiotic and probiotic therapies, with the goal of improving resilience against chronic inflammation,” Talcott said. “Thus, the sales of botanical supplements are expanding despite scarce scientific data to support these remedies’ effectiveness in reducing inflammation.”


